Physiological Healing Procedure and Histological Observation
作者:Rong Xiang Xu 出版社:KARGER 发行日期:In 2004INTRODUCTION
Moist-exposed burn Therapy is a local burns management by which burned tissue is exposed to a three-dimensional physiologically moist environment to reject, regenerate and repair [1]. We herein present a report on the dynamic pathomorphological changes of deep second-degree burns wounds. Studies in this subject are seldom performed and seldom available.
MATERIALS AND METHODS
Clinical data
Six cases were enrolled throughout the study, 5 males and 1 female, aging from 21 to 36 years. Total burns surface area varied from 12% to 45%, with an average of 24.33% 12.37%. All patients were admitted within 6 h postburn. After admission, patients received fluid resuscitation and antibacterial therapy on the basis of burn severity. Burns wounds were applied with MEBO at a thickness of 1mm, which was renewed every 4~6 h till wound healing.
Pathological observation
Tissue specimens were collected on days 1, 3, 5, 7, 10, 15 and 20 postburn and after healing from wound sites including trunk, upper limb and both lower limbs. All specimens were allocated into two parts, one immobilized with 10% formalin, embedded in routine paraffin, sliced up and HE stained for light-microscopic observation; the other was immobilized with 2.5% glutaraldehyde solution, prepared on routine basis for observation using TEM 1200EZX-model electron transmission microscope.
RESULTS
1) Light microscopic observation:
Under the light microscope, all burns wounds were noted to involve deep dermis and the pathological changes of burns wounds at different time phases were almost the same.
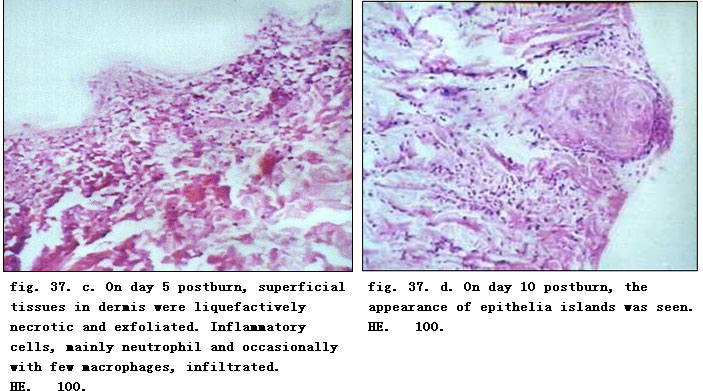
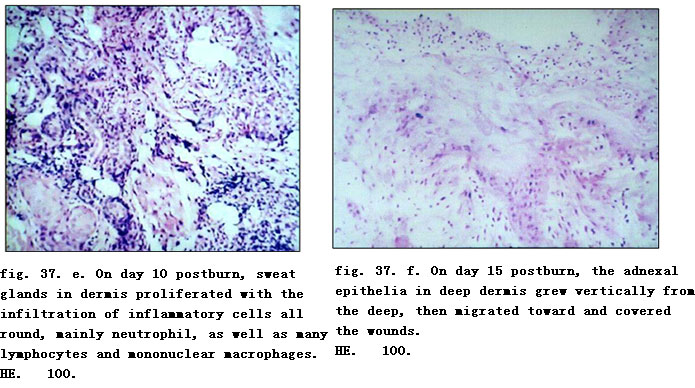
On day one postburn, epidermis became necrotic due to coagulation and necrotic basal layer cells were seen arranged in a column-fencing formation. Edema was present between epidermis and dermis. Collagenous fibers in the superficial layer of dermis were denaturated, and swollen and loosely arranged while epithelia of the dermal adnexal epithelia were also denaturated. We observed contracted nucleoli of the capillary endothelium, blood clot or stasis in lumen with little infiltration of neutrophil (fig. 37. a). Only collagenous fibers and dermal appendages in deep layer of dermis were approximately normal. On day 3 postburn, necrotic epidermal cells progressed to vacuolation with mild edema between the epidermal and dermal layers. Collagenous fibers in the superficial dermis had hyaline degeneration while in the deep tissue, the structure was loose, and blood vessels were slightly swollen with congestion. Scattered inflammatory cells (mainly neutrophils) infiltrated the tissue (fig. 37. b). On day 5 when the necrotic epidermis exfoliated, superficial tissue in the dermis liquefied and loosened due to necrosis and neutrophilic infiltration (occasional lymphocyte and mononuclear macrophages) (fig. 37. c). On day 7 postinjury, the aforementioned pathological changes became more notable. On day 10, the superficial necrotic tissue in the dermis liquefied, discharged and thinned. A large amount of neutrophils infiltrated into the liquefied necrotic tissue, interwoven with few mononuclear macrophages. The dermal adnexal epithelia in the underlying deep tissue revealed squamous metaplasia with enlarged cells, deep karyon, rich and red cytoplasm that clustered to form “epitheium island” (fig. 37. d). Among these islands, fibroblasts proliferated and were characterized by large volume of cells, conspicuous nucleolus (binucleolate was frequently present) and basophilic cytoplasm. In addition, infiltration of many inflammatory cells including neutrophil (mainly), lymphocyte and mononuclear macrophages were visible in wounds (fig. 37. e). On days 15 and 20 postburn, an inflammatory exudation layer replaced the necrotic layer whose liquefaction was now accomplished. The underlying residual adnexal epithelia, fibroblasts and endothelia showed active proliferation. The regenerated “epithelial island” grew vertically, then migrated toward and covered the wounds (fig. 37. f, g). Neoformative capillaries were noticed and granulation tissues were formed. The infiltration of inflammatory cells (mainly lymphocytes) in the dermis was still present, especially in the periphery of the regenerated skin appendages. After healing, regenerated skin appeared to be almost normal in structure and the majority of skin appendages were restored completely. Infiltration of certain inflammatory cells and few macrophages in dermis persisted (fig. 37. h).


2) Electron-microscopic Observation:
Ultrastructure alterations of the superficial dermis layer were observed on days 1, 3 and 5 postburn, while those of full-thickness skin were done on day 7, 10, 15 and 20 postburn and after healing.
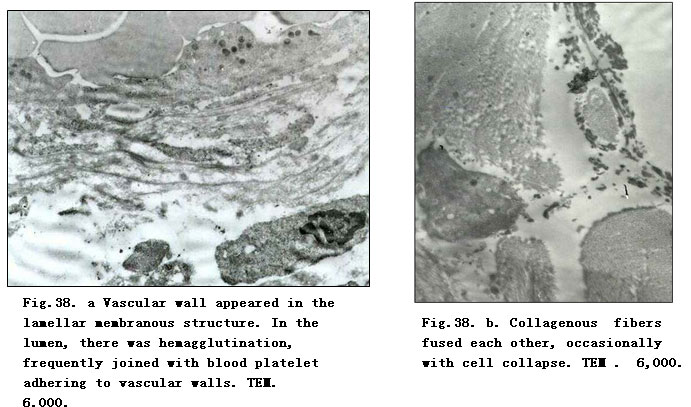
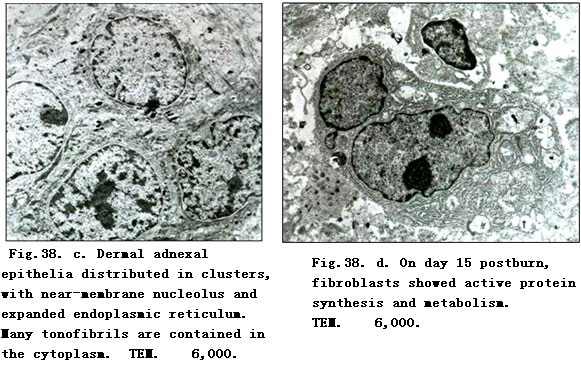


On day 1 postburn, electron-microscopic observation indicated loose and disorderly arranged collagenous fibers in the superficial dermis, and a distinct and recognizable light-dark zone of the fasciculus. Fibroblasts which had a cytomembrane profile which remained the same although being deprived of the cell organ. Nuclear membrane loomed with increscent nucleopore to cause the interpolation of nuclein and chromatin, leading to lumpish chromatin. Microangial endothelia had similar changes as did the fibroblasts. The vascular walls appeared as a lamellar membranous structure. In the lumen, we noted hemagglutination, frequently joined with blood platelet adherence to the vascular walls (Fig.38. a). On day 3 postburn, collagenous fiber bundles in the superficial dermis varied in thickness and appeared disorderly with faint or absent light-dark zones. The cell ultrastructure interwoven between fibers disappeared. On day 5 postburn, the superficial collagenous fibers merged to produce floccules with high electron density surrounded by scattered electro-dense organelle-like structures and necrotic cell fragments as well as occasionally by varied lower electodense lipid droplets without coating of the limiting membrane (Fig.38. b). On day 7, when the ultrastructure change of the superficial dermis was similar to that on day 5, deep collagenous fibers and the structure of dermal adnexal epithelia nearly returned to normal, except for a slight broadening of the intercellular space. On day 10 postburn, fibroblasts in the dermis showed condensed nuclear chromatin, hypertrophic and shift aside nucleoli. Rough endoplasmic reticulum (RER) proliferated showing flat vesicular and vesicular-like expansion, while residual dermal adnexal epithelia were distributed in clusters containing a high amount of tonofibril (Fig.38. c). On days 15 and 20 postburn, collagenous fiber bundles of dermis varied in thickness and appeared disorderly; fibroblasts were binucleated and showed obvious nucleoli with the presence of karyosome; perinuclear pool demonstrated beading swelling and the RER proliferated (Fig.38. d). In dermal adnexal epithelia, nucleoli were distinct and increased numbers of karyosomes were frequently observed.
The chondriosome was rich and intracytoplasmic tonofibril showed an increase in cytoplasmic presence. Also, we observed a gradual establishment and improvement of the intercellular desmosomal junction. After healing, neoformative epidermis cells and intercellular junction recovered almost to normal and the integrated dermis-epidermis junction reappeared (Fig.38. e). The majority fibroblasts in the dermis appeared as long strip rich in cytoplasm with well-developed organelles. However, no additional active metabolism of the same was observed. Fibroblasts were uniform in thickness, fasciculated and well orientated (Fig.38. f). The healing skin of deep second-degree wounds became almost identical to that of noninjured skin (Fig.38. g).
CONCLUSION
Pathological changes of the deep second-degree burn wounds treated with BRT with MEBT/MEBO can be divided into three stages: 1) denaturation and necrosis, 2) liquefaction, and 3) restoration. These stages may overlap. Burns wounds healed by complete physiological regeneration.
DISCUSSION
Among the available literatures regarding the pathological changes of burns wounds treated with BRT with MEBT/MEBO, the majority report the light microscopic observation before and after treatment with MEBT [2-4]. There are relatively few monographs or systemic study reports on the ultrapathologic processes during BRT with MEBT/MEBO.
Our study demonstrated that the pathological changes of burns wounds treated with BRT with MEBT/MEBO were totally different from pathological processes when treated conventionally [5, 6]. On days 1~3 postburn, the so-called “acute inflammatory response period” with its typical denaturation and necrosis occurred in the burned epidermis and superficial dermis, revealing a slight inflammatory response, but without signs of “leukocyte infiltration zone”. Then the necrotic tissue began to liquefy and discharge increasingly as the disease course progressed. Such changes climaxed around day 10 when dermal adnexal epithelial cells, fibroblasts as well as other repaired cells showed signs of regeneration. Microscopic observation indicated large cell bodies of fibroblasts, increased basophil of the cytoplasm, hypertrophic nucleoli,and RER which proliferated, demonstrating the typical flat vesicular and vesicular-like expansion. At this time, the proliferation and migration of residual dermal adnexal epithelia and proliferation of granulation tissues became dominant. The most spectacular observation was that the “epithelia island” (formed by the regenerated adnexal epithelia) initially grew vertically, then migrated toward and covered the wound as granulation tissues proliferated. This resulted in a flat skin surface. During this period, either residual adnexal epithelia or fibroblasts displayed “active protein synthesis and metabolism”[7]. The prominent characteristic changes involved significant proliferation of RER and the appearance of increased karyosomes. Finally, the wounds were noted to be completely covered by regenerated squamous epithelia as healing was accomplished.
We observed that inflammatory cells demonstrated a series of responses during wound healing. On day 1 postburn, we observed infiltration of neutrophils into the dermis followed by lymphocytes and mononuclear macrophages. This infiltration increased on day 3. Neutrophils were the predominant infiltrating cells in necrotic tissue, and their numbers grew or declined depending upon the length of liquefaction time. Meanwhile, lymphocytes and mononuclear macrophages, mainly located in the residual dermis, dramatically increased during the period of time that necrotic tissue was being rejected and wound repair was initiated. These cells remained the dominant cells impacting wound repair subsequent to the liquefaction period. This study does not resolve questions as regards the significance of the space-time distribution of these cells nor does it answer definitively questions about sequential changes. These questions merit further study. However, we do note the remarkable truth that, during wound healing, the function and activity of the above cells directly or indirectly, alone or cooperatively, participated in and regulated wound healing.
The study results indicated a staged pathological change of burns wounds when treated with BRT with MEBT/MEBO. Wounds changed in three stages according to the different time phase postburn: (1) denaturation and necrosis stage; (2) liquefaction stage, and (3) stage of repair by regeneration, which may overlap during wound healing.
The first stage began subsequent to burn injury till day 3 and the pathomorphological change was characterized by the denaturation and necrosis of burns tissue from which we derived the name “denaturation and necrosis stage”. We postulated that these changes were the result of direct thermal exposure, local microcirculation blockage and other secondary injuries. The minor inflammatory response at this stage had a close correlation with the effects of MEBT/MEBO. On day 5 postburn, wound liquefaction became conspicuous and climaxed on day 10. After this time, liquefaction diminished and we noted less necrotic tissue. The inflammatory response, however, remained correlated with the liquefaction of necrotic tissue. Repair by regeneration was noted on day 10 postburn and was manifested mainly by active proliferation of residual dermal adnexal epithelia. This stimulated the proliferation of peripheral fibroblasts and endothelia, and further formed granulation tissues, leading to final healing by epithelization. After experiencing further differentiation and reformation, the regenerated skin finally attained the structure of normal skin. This study demonstrates that the effect of BRT with MEBT/MEBO delivered similar pathological changes for both deep second-degree and third-degree burns wounds. The only difference lay in the ultimate healing modes.
Remarkably, this study revealed that the healed wounds appeared as flat fully regenerated skin, featuring restored, viable hair, almost normal skin elasticity and no scar formation. Histomorphological observation confirmed that the epidermal cells of neoformative skin had the equivalent structure as normal epidermis and that the epidermis had good joints with dermis papilla. The healed skin demonstrated a fully functional and integrated dermis-epidermis junction. The initially infantile and active fibroblasts gradually grew into stable fibrocytes. Arteriolae and venulae with thick walls and integrated structure replaced the neoformative capillaries. Collagenous fibers were uniform in thickness and orientated fascicularly, without the presence of whirl-like and nodular arrangement. Follicles, sweat glands and sebaceous glands as well as other dermal accessory organs also regenerated completely. Therefore, we concluded that treatment of deep second-degree burns wounds with BRT with MEBT/MEBO results in complete physiological regeneration with minimal scar formation.
REFERENCES
1. Xu RX: Brief Introduction on Chinese moist medical science for burns. Chin J Burns Wounds Surface Ulcers 1996, 8: 1-6.
2. Wang GS, Jian WG: Pathological Changes of rabbit with experimental burns treated by MEBO and investigation of the mechanism of action. Chin J Burns Wounds Surface Ulcers 1992, 4: 7-11.
3. Zhang XZ, Cheng SR: A case report: Electron microscopic observation on burn wound treated with MEBO. Chin J Burns Wounds Surface Ulcers 1992, 4: 6-9.
4. Zhao YL, Wang Y: Electron microscopic observation on the effect of MEBO in treating deep burn wound. Chin J Burns Wounds Surface Ulcers 1995, 7: 1-5.
5. Fang ZY, Wu ZL: Burn Theory and Practice. Shenyang, Liaoning Science and Technology Publishing House, 1989, pp 12-14.
6. Fu XB, Wang DW: Basis of Trauma Repairing. Beijing, People’s Military Medical Press, 1997, pp 14-30.
7. Wu ZB: Basis of Ultra-Micro-Pathology. Beijing, People’s Health Press, 1990.