Primary Exploration on the Mechanism of the Anti-Infection E
作者:Rong Xiang Xu 出版社:KARGER 发行日期:In 2004INTRODUCTION
Clinical practice has revealed that MEBO has a remarkable anti-infection effect [1]. During the period May to June 1992, fourteen burns cases were treated in the Burns Department, Affiliated Hospital of this college, and bacteria isolated from the burns wounds were examined. We found that Bacillus proteus had the morphological Hauch-ohne Hauch (H-O) variation, and the plasma coagulation ability of S. aureus decreased. In order to investigate the mechanism of the anti-infection effect of MEBO, we studied the biological variability of some common bacteria, such as Bacillus proteus, P. aeruginosa, E. coli and S. aureus, cultured in medium containing a certain amount of MEBO. The effect of MEBO on non-specific immunity in vivo was also observed.
MATERIALS AND METHODS
Clinical data
During the period from May to June 1992, 14 cases of burns were treated with MEBO (hospitalized for 4 - 20 days). Swab samples were taken from the upper and lower (contact with wound) layers of the MEBO ointment, before changing the dressing. Bacteria were isolated and cultured.
Reagents, Bacterium Species and Culture
Antibiotic sensitivity test paper and nutrient agar were purchased from Shanghai Medical Chemistry Institute. B. proteus, P. aeruginosa, E. coli and S. aureus were prepared in our department.
The four above bacteria were cultured on ordinary culture medium and medium containing different concentrations of MEBO, respectively, and continuously transferred to 10~15 generations. Each generation of the bacteria was checked. The biological characteristic and drug sensitivity of bacteria were examined.
Animal Experiment
Forty healthy adult mice of both sexes weighing 20~24 g were randomly divided into 3 groups, i.e. blank control group (group 1), liquid paraffin control group (group 2) and MEBO group (group 3). Animals in groups 2 and 3 were depilated (2.5 2.5 cm) on their backs and liquid paraffin or MEBO was applied on the depilated area. The frequency was twice a day for 10 successive days. On the 11th day, the mice were sacrificed and abdominal cavity fluids were sampled 30 min after intraperitoneal injection of 0.5 ml of 2% sheep erythrocytes.
Observation Indexes
Bacteria Variation. The four bacteria were cultured and transformed, 18 - 24 h as one generation. The dynamics of every generation of the bacteria was observed under a dark-field microscope. The bacteria were stained with the G method to observe the staining reaction and morphological characteristic. The colony features, biochemical reactions, and the ability of plasma coagulation were also examined.
Nucleoplasm Staining. The 7th generation of B. proteus was smeared, fixed with the vapor of 1% molybdic acid solution, hydrolyzed DNA with 1% hydrochloric acid for 15 min, then stained with Löffler’s methylene blue and observed.
Antibiotic Drug Sensitivity Test: The four bacteria were cultured on ordinary agar medium and medium containing 20 % of MEBO, respectively. Drug sensitivity test paper of eight kinds of antibiotics, i.e. gentamycin, neomycin, erythromycin, carbenicillin, ampicillin, kanamycin, chloromycetin and polymyxin, were stuck on the medium, respectively, and observed after 24 h. The diameter of the bacterial inhibition zone was measured [2].
Determination of Phagocytic Function of the Intraperitoneal Phagocytes. The abdominal cavity fluids were smeared and stained with Wright’s method. The percentage of phagocytes was determined [3].
Determination of Lysozyme Activity in Abdominal Cavity Fluid and Serum. The agar plate method was used: 2 ml of Micrococcus solution (91010/ml) were added to 1 % agar at 70oC, then after mixing well, this was poured into a Petri dish. After cooling, holes (3 mm in diameter) were bored into the substance. Mice serum from 3 groups was added to the holes in one set of Petri dishes and abdominal cavity fluid was added to the holes in another set of Petri dishes. These were incubated at 37oC for 24 h. The diameter of the bacteriolytic ring was measured [4].
Total Number and Classification of Leukocytes in Pperipheral Blood. Blood samples from the tails were taken from 3 groups of mice. Leukocytes were counted, stained with Wright’s method and classification was determined.
RESULTS
1) Species and Variation of Bacteria Isolated from Burns Wounds
Table 42 shows that after treatment with MEBO, B. proteus in the wounds had H-O morphological variation. S. aureus in the wounds turned from positive plasma coagulase to negative, or the plasma coagulating ability decreased.
2) Bacteriostasis of MEBO
Eight species of bacteria, i.e., S. aureus, S. albus, E. Coli, B. proteus, P. aeruginosa, B. typhosus, B. paratyphoid A and B. dysenteriae, were cultured in simple agar dishes. The scraps of MEBO filter paper were pasted on the bacterial surface of streak plating. The results showed that MEBO had no direct bacteriostasis or bactericidal action. 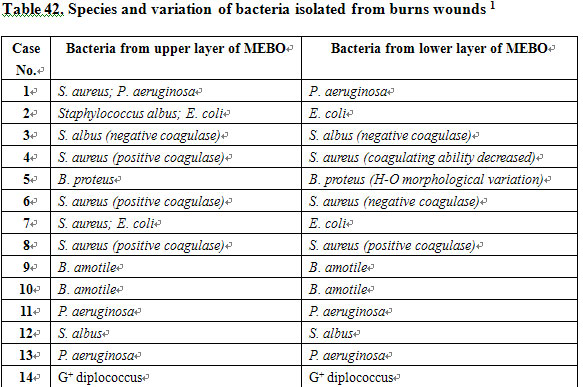
3) Effect of MEBO on Bacterial Biological Characteristics
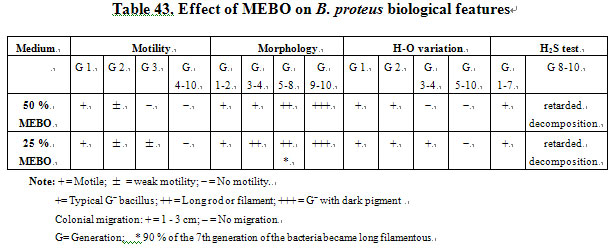
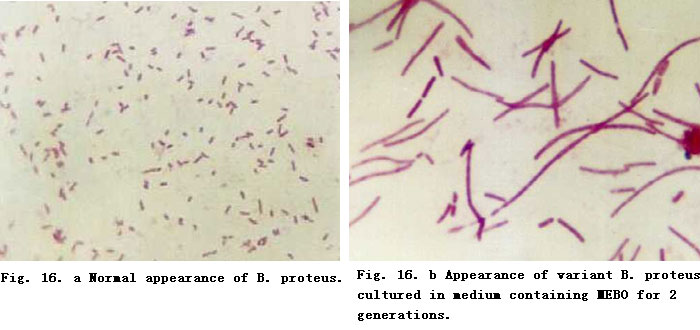
Effect of MEBO on B. proteus Biological Features. B. proteus was cultured for several generations on medium containing certain amounts of MEBO. We noted that
the motility of the bacteria gradually decreased before finally vanishing. Also, we noted that H-O variation occurred. The 7th generation of the bacteria
became long and filamentous. In culture medium containing 25 % MEBO, 90 % of the bacteria became long filamentous or long rod, and then became small
bacillus. Dark pigments appeared, colonies became small and the bacteria grew very slowly. The decomposition activity of the bacteria to glucose and lactic
acid was retarded (Table 43; Fig. 15). The effect of MEBO on H antigen of B. proteus is shown in Table 44.

Effect of MEBO on the Biological Characteristic of P. aeruginosa. It was found that P. aeruginosa cultured in MEBO-containing medium started to decrease its motility from the 5th generation, and motility vanished in the 10th generation. Variations of morphology and colonial features of the bacteria were also found (Table 45).

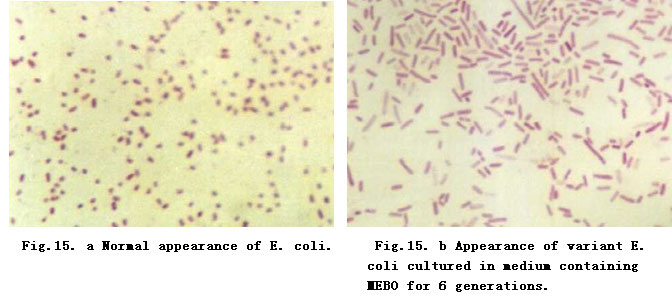
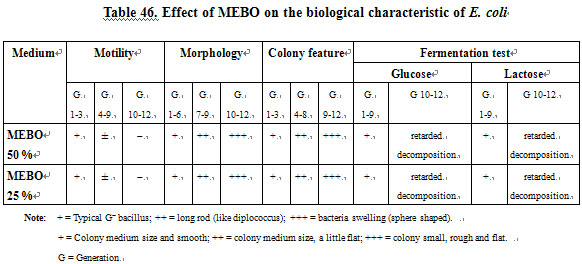
Effect of MEBO on the Biological Characteristics of E. coli. From Table 46, we can see that after proliferation to the 10th generation while cultured in MEBO containing medium, E. coli changed as follows: It lost motility, became sphere shaped, colonies became smaller, dry and flat (also Figure 16). The decomposition activity of the bacteria to glucose and lactose was retarded (after 32 h).
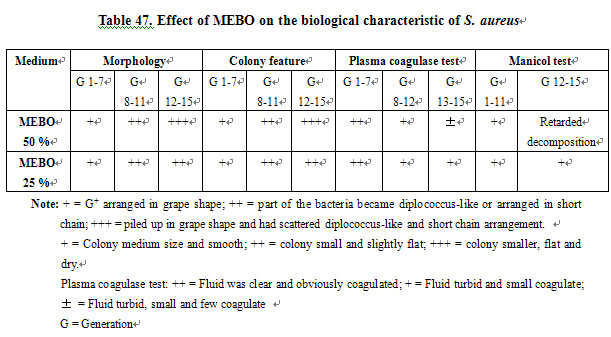
Effect of MEBO on the Biological Characteristic of S. aureus. We found that after 10 generations of S. aureus cultured in medium containing MEBO, bacteria were piled up in grape shape and had scattered diplococcus-like and short chains. Colonies became smaller, flat and dry. The decomposition activity of the bacteria to manitol was retarded (after 32 h), and the variation of plasma coagulation ability was very significant (Table 47) [3].
Nucleoplasm Staining of B. proteus. Cultured in medium containing 25 % MEBO and proliferated to the 7th generation, B. proteus appeared as a long filamentous variant. Nucleoplasm staining was done and examined. As the RNA in cytoplasm was hydrolyzed, the nucleoplasm was stained blue. The bacteria became long rod or long filament in the course of binary division, because the formation of cell wall was slower than the division of the nucleoplasm. This result proved that bacteria proliferation was retarded when cultured in medium containing MEBO.
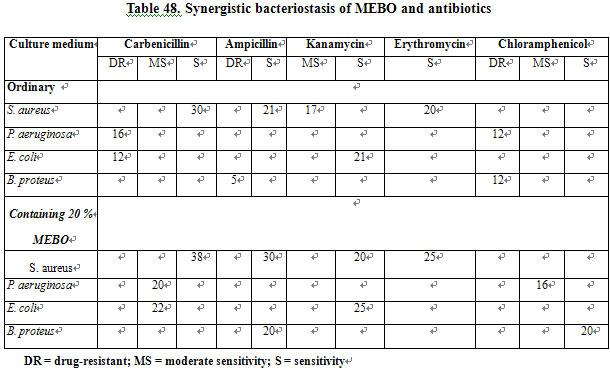
Synergistic Effect of MEBO and Antibiotics on Bacteriostasis. When cultured in medium containing 20 % MEBO, P. aeruginosa resistant to carbenicillin and chloromycetin became moderately sensitive and B. proteus resistant to chloromycetin and ampicillin became sensitive. Both carbenicillin and kanamycin had a synergistic effect with MEBO against E. coli. Carbenicillin, ampicillin, kanamycin and erythromycin had a synergistic effect with MEBO against S. aureus (Table 48).
4) Effect of MEBO on Nonspecific Immunity
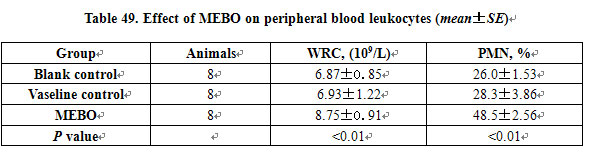
Effect on Peripheral Blood Leukocytes. MEBO significantly increased the peripheral blood count of white blood cells and polymorphonuclear leukocyte (PMN %) in mice (Table 49).
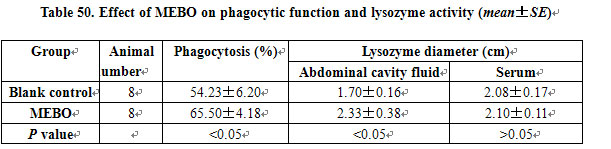
Effect on Phagocytic Function and Lysozyme Activity: MEBO significantly promoted the phagocytic function of phagocytes and increased the lysozyme activity in the abdominal cavity fluid (Table 50).
CONCLUSION
MEBO can increase the number of WBC in the peripheral blood of mice. It can also enhance the phagocytic function of phagocytes in the abdominal cavity of mice. MEBO can induce a variation of bacteria and improve nonspecific immunity.
DISCUSSION
Bacterial inhibitory test proved that MEBO does not have a direct bacteriostatic or bacteriocidal effect. This finding may be understood in part due to oily texture of MEBO making it very difficult to infiltrate and diffuse into a watery culture medium. After continuous culture in medium containing MEBO, many species of bacteria showed variations in morphological structure and biological characteristics that are closely related with the growth environment. The morphological variation of bacteria may cause changes in its biochemical characteristics, antigenicity and toxicity. B. proteus and P. aeruginosa had deep pigmented particles and E. coli became sphere-shaped after cultured in medium containing MEBO.
These variations are non-genetic. MEBO had a synergistic bacteriostatic effect with antibiotics. This is beneficial to the control of local and systemic infections secondary to severe burns. MEBO promoted the phagocytic function of abdominal cavity phagocytes and release of lysozyme, and increased the leukocyte and neutrophil counts in the peripheral blood. This is very important for clearing out the bacteria and toxins both locally and systemically. In summary, the mechanism of the anti-infective effect of MEBO includes inducing variation of the bacteria, decreasing their proliferation rate, reducing bacterial pathogenicity and promoting non-specific immunity of the body.
REFERENCES
1. Zhang LX, Yang KF: Clinical report of 2076 burn cases treated with moist exposed burn therapy. Chin J Burns Wounds Surface Ulcers 1989; 1: 22-26.
2. Shanghai Medical Laboratory: Test of Sensitivity to Antibiotics. 1983.
3. Yu H (ed): Medical Microbiology. Beijing, People’s Health Publishing House, 1983, vol 13-28,p 239.
4. Wang MX (ed): Medical Microbiology and Immunology. Beijing, People’s Health Publishing House, 1989, p 116.